In the fast-evolving aesthetic device industry, the 2940nm Er:YAG laser has become one of the most respected technologies for precision skin resurfacing, scar improvement, wrinkle reduction, and overall rejuvenation. What makes SANHE Laser especially noteworthy in this segment is not only the product itself, but also the company’s long development background in medical-aesthetic laser equipment. SANHE was established in 2005, and its history page shows that the company had already been expanding its energy-based device portfolio by 2010 and 2011. Based on that timeline, it is reasonable to describe SANHE as an early participant in China’s aesthetic laser manufacturing field, which helps explain why its 2940nm Er:YAG system is presented today as a mature and clinically oriented platform rather than a late-following product.
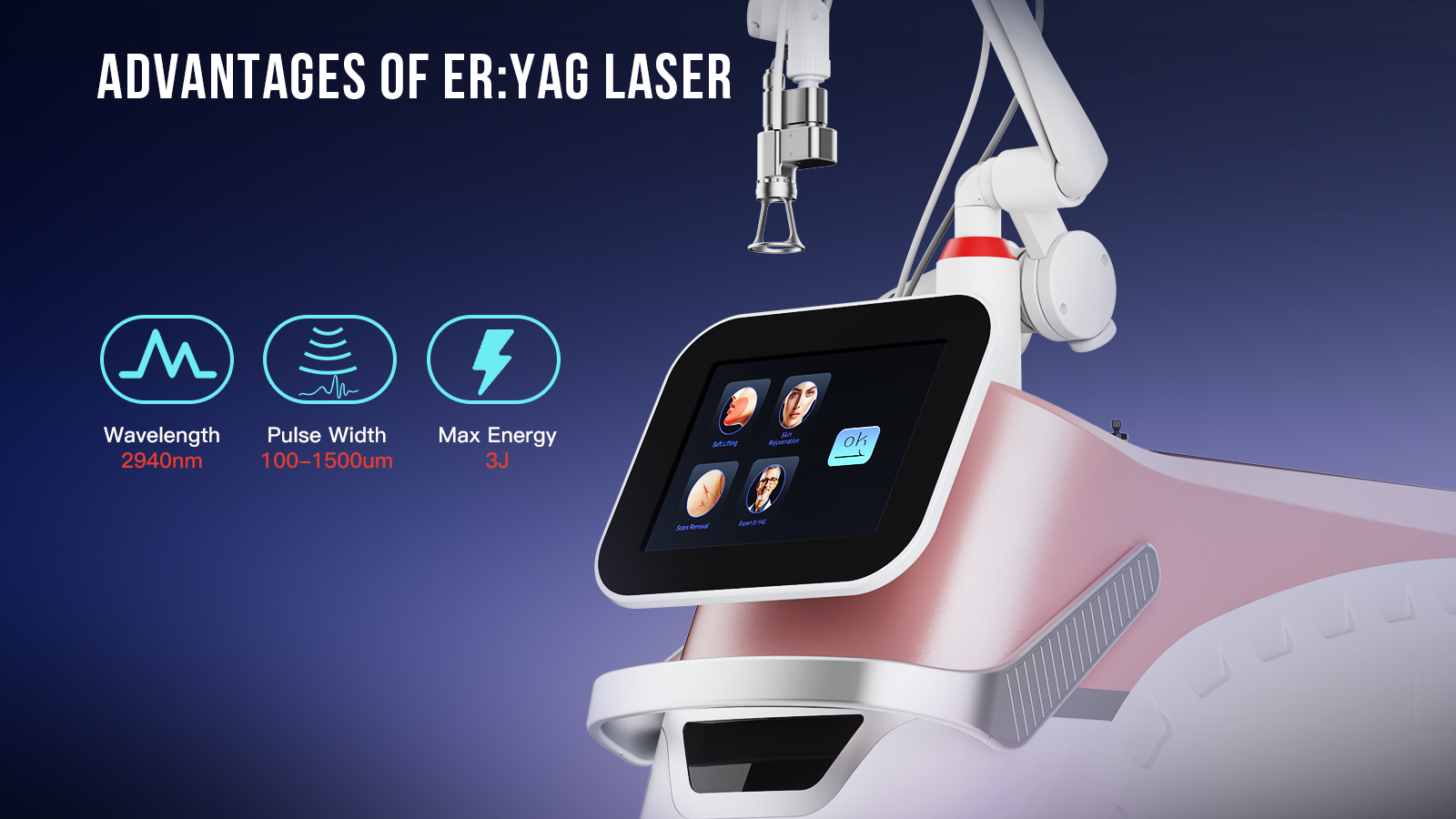
From a technology standpoint, SANHE positions its 2940nm Erbium Yag Laser machine as a high-precision system designed for skin rejuvenation, wrinkle reduction, pigment treatment, scar improvement, eyelid-area applications, and resurfacing with limited downtime. On the product page, the machine is described as offering micrometer-level evaporation control, minimal thermal damage to surrounding tissue, reduced pain, and fast recovery. The interface also includes guided parameter selection and the ability to save treatment settings, which are important features for clinics seeking both treatment consistency and easier operator training. These details suggest a platform developed with practical clinical workflow in mind, not just headline specifications.

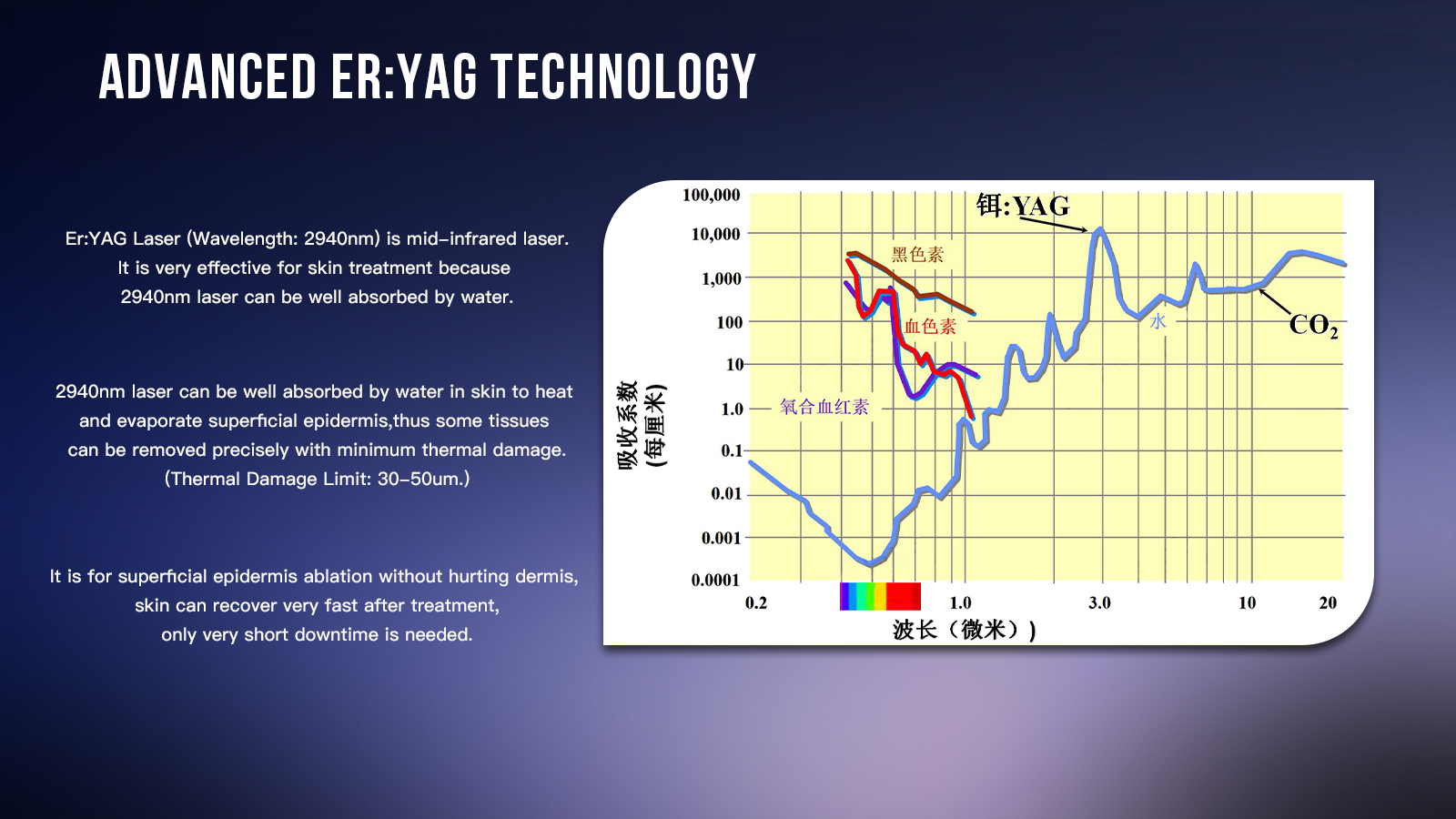
The clinical relevance of the 2940nm wavelength is also well supported in the broader medical literature. StatPearls notes that Er:YAG lasers emit at 2,940nm, a wavelength strongly absorbed by water in the epidermis and dermis, making them effective for resurfacing and collagen-related rejuvenation. Published clinical studies also support the category’s treatment value: a 2020 PubMed study on acne scars reported progressive improvement across repeated fractional ablative Er:YAG sessions, while a prospective study on periocular rejuvenation found statistically and clinically significant improvement after three sessions, with only mild short-term erythema and edema and no long-term adverse effects reported. In other words, the treatment logic promoted on SANHE’s product page is aligned with established evidence around 2940nm Er:YAG technology.
What strengthens SANHE Laser’s reputation further is the company’s broader manufacturing and service structure. SANHE says it has its own R&D department, clinic, sales, and after-sales departments, and that it can provide technical support and clinic data quickly. Its company materials also emphasize quality control, customized OEM/ODM capability, operator training, and ongoing machine support. In the FAQ, SANHE states that products are inspected at every stage of manufacturing and again as a complete unit before shipping, and that it maintains an after-sales team to resolve customer issues. For distributors and clinics, these are exactly the factors that shape positive feedback over time: stable product quality, responsive support, and confidence that the supplier will still be present after installation.

This is why SANHE Laser’s 2940nm Er:YAG machine can be presented as more than just another resurfacing device. It reflects a company with a long development track record, an established manufacturing base, and a product concept that matches the core strengths of Er:YAG technology: precision, controlled ablation, limited collateral heat, and clinically relevant rejuvenation outcomes. Even more importantly, the brand’s positioning around R&D, clinic support, quality inspection, and after-sales service helps explain why the product has earned a favorable market image among many professional buyers looking for dependable long-term cooperation, not just a low-cost machine.
.jpg)
A small accuracy note: I found strong support for SANHE’s company history, product positioning, inspection process, and after-sales claims, plus strong third-party evidence for the 2940nm Erbium Yag Laser machine category itself. I did not find a peer-reviewed, SANHE-specific published clinical trial on that product page, so I kept the clinical wording precise rather than overstating it.